Spine
 Back pain affects eight out of ten adults during their lifetime. It has a huge economic burden on society, estimated to cost as much as 1-2% of gross national product in several European countries. Treatments for back pain have lower levels of success and are not as technologically mature as those for other musculoskeletal disorders such as hip and knee replacement. Our research focusses on the development and evaluation of treatments for disc degeneration, vertebral fracture and spinal cord injury.
Back pain affects eight out of ten adults during their lifetime. It has a huge economic burden on society, estimated to cost as much as 1-2% of gross national product in several European countries. Treatments for back pain have lower levels of success and are not as technologically mature as those for other musculoskeletal disorders such as hip and knee replacement. Our research focusses on the development and evaluation of treatments for disc degeneration, vertebral fracture and spinal cord injury.
Vertebral fracture and repair
There are estimated to be 1.4 million new osteoporotic vertebral fractures each year worldwide. A minimally invasive treatment called vertebroplasty has been introduced where a special cement is injected into the damaged vertebra to stabilise the fracture and reduce the pain. However, the treatment is controversial with large variations in the outcomes. If we could gain a better understanding of which patients would benefit biomechanically, then the treatment could be better targeted.
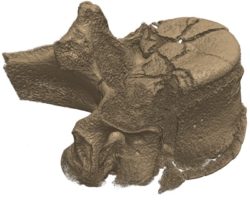 We have developed methods for evaluating the biomechanical performance of vertebroplasty using experimental in vitro testing and computational modelling. We have worked with biomaterials research groups to evaluate and optimise cement properties, and are developing techniques to simulate the performance of the treatment over longer periods of time. We are also evaluating the efficacy across a patient population, using computational models that represent the variation in anatomy and properties seen clinically.
We have developed methods for evaluating the biomechanical performance of vertebroplasty using experimental in vitro testing and computational modelling. We have worked with biomaterials research groups to evaluate and optimise cement properties, and are developing techniques to simulate the performance of the treatment over longer periods of time. We are also evaluating the efficacy across a patient population, using computational models that represent the variation in anatomy and properties seen clinically.
If you'd like to hear more, please contact:
Treatments for the intervertebral disc
Low back pain is strongly associated with degeneration of the intervertebral discs, the soft tissues that connect the spinal vertebrae and allow the spine to articulate. Current surgical treatments for low back pain are highly invasive and have relatively low long term success rates.
Through two major grants, we are developing novel therapies for nucleus and disc replacement and methods for evaluating and optimising their performance pre-clinically. These include novel biomaterials for nucleus augmentation, combination therapies including high intensity focussed ultrasound (in collaboration  with Oxford), and acellular disc scaffolds. We have developed new pre-clinical testing methodologies including both in vitro and in silico methods, and novel imaging approaches to evaluate the performance of these treatments.
with Oxford), and acellular disc scaffolds. We have developed new pre-clinical testing methodologies including both in vitro and in silico methods, and novel imaging approaches to evaluate the performance of these treatments.
If you'd like to hear more, please contact:
Tissue characterisation
Fundamental tissue characterisation work is required to underpin the development of representative models of the spine as well as to understand key mechanical factors contributing to damage onset and propagation in the tissue.
As the natural tissues have a mechanical behaviour that is highly dependent on their shape and interconnection, we have developed methods that allow to derive their properties while minimally disrupting their in-situ characteristics. This combines mechanical testing, imaging and computational models with reverse engineering to be able to extract key variables that are sometimes not otherwise directly measurable.
We have done this for diverse tissues in the spine (bone, intervertebral disc and ligaments), assessing their physiological function and their damage behaviour when over-loaded.
If you'd like to hear more, please contact:
Spinal Cord Injury (SCI)
Somewhere between 250, 000 and 500, 000 individuals experience a spinal cord injury globally every year, according to the World Health Organisation (WHO), with up to 90% of the cases experiencing traumatic SCI resulting from falls or road traffic injuries. The severity of SCI depends on the level of the spine affected, the injury type, and the intensity of the injury. The consequences are often devastating for the patient and include permanent locomotor and sensory deficits, which may lead to pain, paralysis, urinary and respiratory dysfunction, metabolic problems as well as psychological problems.
Currently treatments focus on the symptoms and presently there is not a cure. Treatment is the same for all patients regardless of the type of injury or the severity of the symptoms. Whilst in the past it was thought that there was no regenerative potential of the central nervous system, it is now recognised that this is not the case and regeneration of neurons in the spinal cord and partial recovery from injury has been reported. However, little is understood about the process of recovery.
Our research is developing 3D models of spinal cord injury by combining biomaterials (e.g. hydrogels) with neural cells and applying injury inducing forces to investigate the effect of altering injury speed, displacement and type. In addition, we are also investigating how altering the stiffness of the extracellular matrix (hydrogel) affects the different cell types of the spinal cord and are currently developing an in vitro model of non traumatic spinal cord injury.
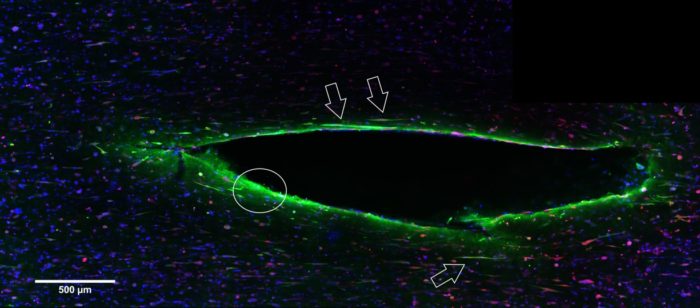
Hydrogel containing spinal cord cells ten days after injury.
The injured gel was imaged in the middle of the injury zone. Cells labelled with GFAP (glial fibrillary acidic protein in green), a marker of activation show co-location with the injury. Cell nuclei are stained blue and dead cells are stained red.
If you'd like to hear more, please contact Professor Joanne Tipper
